Poster P454
P454
Confirmation of germ-free health status in embryo transfer rederived mouse models
Vargas, Andrea1; acevedo, Oscar1; Chavarria, Tony1; Umana, Elmer1; du, gong1; Jordan, Ellen B.1; Varian, Bernard1; Shin, Heaji2; Yilmaz, Omer3; Metcalf Pate, Kelly A.1; Erdman, Susan1; Fox, James G.1
1. Division of Comparative Medicine, Massachusetts Institute of Technology, Cambridge, MA, MA, United States.
2. David H Koch Institute for Integrative Cancer Research, Massachusetts Institute of Technology, Cambridge, MA, United States.
3. Department of Biology, Massachusetts Institute of Technology, Cambridge, MA, United States.
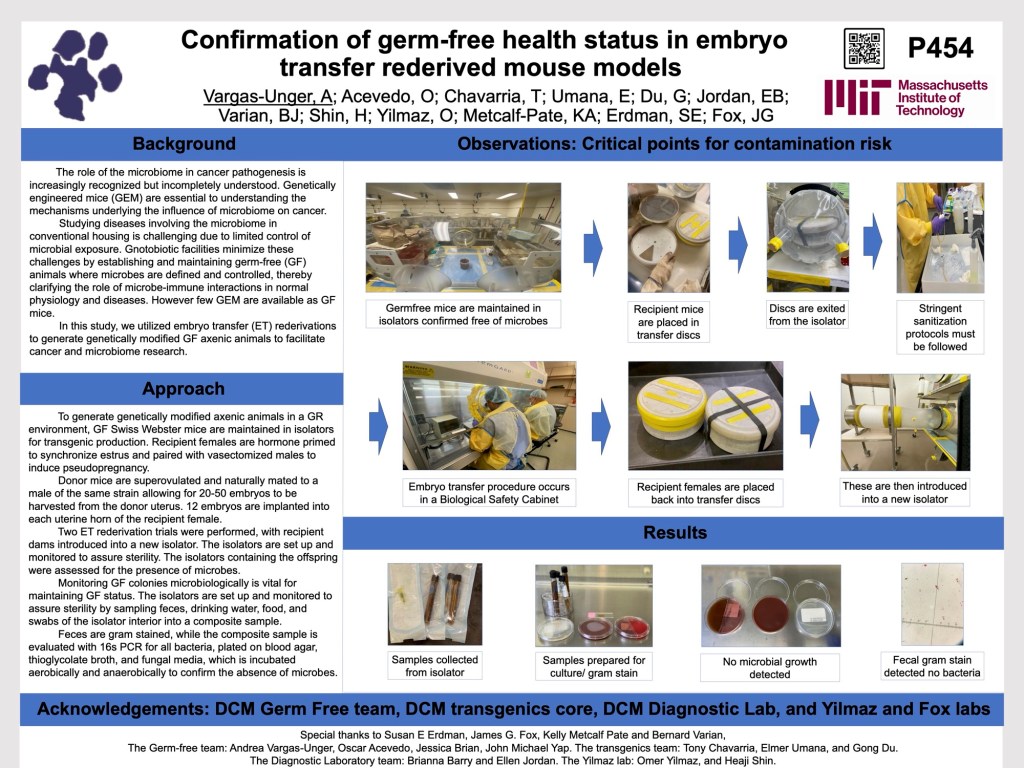
Studying diseases involving the microbiome in conventional housing is challenging due to limited control of microbial exposure. Gnotobiology minimizes these challenges by establishing and maintaining germ-free (GF) animals where microbes are defined and controlled, thereby clarifying the role of microbe-immune interactions in normal physiology and diseases. In this study, we utilized embryo transfer rederivations to generate genetically modified GF anexic animals to facilitate cancer and microbiome research. GF Swiss Webster mice are maintained in isolators for transgenic production. Recipient females are hormone primed to synchronize estrus and paired with vasectomized males to induce pseudopregnancy. Donor mice are superovulated and naturally mated to a male of the same strain allowing for 20-50 embryos to be harvested from the donor uterus. 12 embryos are implanted into each uterine horn of the recipient female. Two ET trials were performed. In one trial, 2 of 4 recipients yielded 8 pups, four of each sex. In the second trial, 1 of four recipients yielded 7 pups, 2 females and 5 males. This ET rederivation presents two critical points for contamination risk. First, highly trained personnel must follow strict protocols. Second, GF recipient females exit an isolator, are transported to a class II BSC with an integrated surgical microscope where donor embryos are implanted, and upon recovery are returned to a new isolator. Monitoring GF colonies microbiologically is vital for maintaining GF status. The isolators are set up and monitored to assure sterility. Isolators are confirmed free of microbes by sampling feces, drinking water, food, and swabs of the isolator interior into a composite sample. Feces are gram stained, while the composite sample is evaluated with 16s PCR for all bacteria, plated on blood agar, thioglycolate broth, and fungal media, which is incubated aerobically and anaerobically to confirm the abscense of microbes. The isolators containing the offspring generated from the ET rederivation trials were confirmed GF with no aerobes, anaerobes, or fungi present, no bacteria on fecal gram stain, and 16s PCR negative.